Ernest Rutherford
|
R.V:Ernest Rutherford a Chemist and physicist.he was born August 30, 1871, in Spring Grove, New Zealand and he died in Cambridge, England, on October 19, 1937 of a strangulated hernia. He was pioneer of nuclear physics and was the first person to split the atom. Rutherford was awarded the 1908 Nobel Prize in Chemistry for his theory of atomic structure and also known as “Father of the Nuclear Age”.
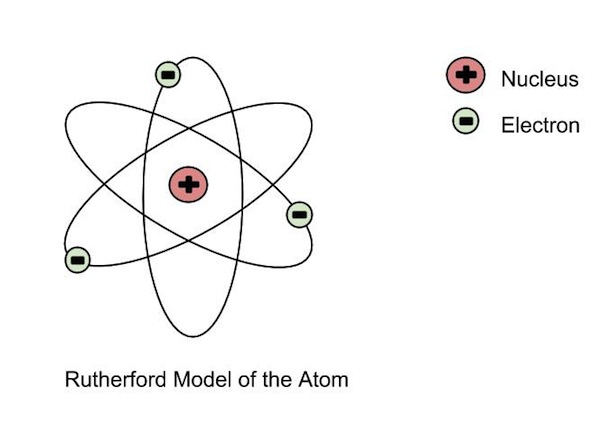
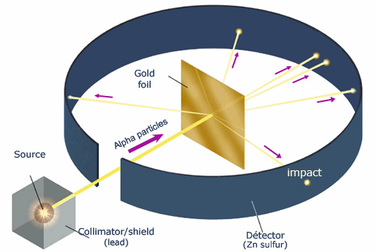
Ernest Rutherford had three major discoveries that shaped modern science, created nuclear physics and changed the way that we predict the structure of the atom.Rutherford first discovered that elements are not unchangeable, but can change their structure naturally, from heavy elements to slightly lighter.This led to him being awarded the Nobel Prize for Chemistry in 1908, for his work on the transmutation of elements and the chemistry of radioactive material.His second discovery, was of the nuclear model of the atom.The model became the basis for how we see the atom today.Through many experiments such as where he bombarded gold foil with alpha particles.The atom model consists of a tiny nucleus surrounded by orbiting electrons in any any ways.He built on this discovery for his third achievement which was the splitting of the atom.He also found out that the atom is 99.99% empty space, the nucleus is positively charged and contains most of the mass of the atom and that the nucleus is about 100,000 times smaller than the atom itself. Rutherford’s model differs to J.J. Thomson's model of the atom.This is because unlike Thomson's model of a atom was a sphere of positive matter in which electrons are positioned and embedded , Rutherford’s model of a atom is composed of a very small and dense nucleus, with orbiting electrons making up most of the atom's volume. |
M.F:It was his interpretation of these experiments in 1911 that led him to the Rutherford model of the atom, involving a very small positively-charged nucleus orbited by even tinier negatively-charged electrons, a great advance on J. J. Thomson’s so-called “plum pudding” model. Ernest Rutherford performed an experiment to test the plum pudding model. He fired energetic a (He2+) particles at a foil, and measured the deflection of the particles as they came out the other side. From this he could deduce information about the structure of the foil. |