Niels Bohr
R.V:Niels Bohr was a Danish physicist and a student of Rutherford’s. he was born on October 7, 1885, in Copenhagen, Denmark and after having a stroke, he died on November 18, 1962, in Copenhagen.He made a revolutionary theory on understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922.
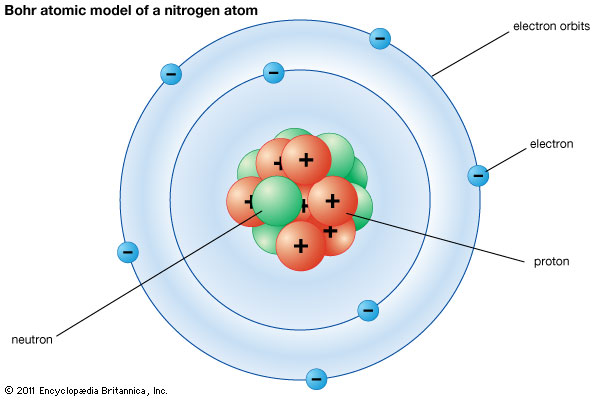
Through discoveries of the electron and radioactivity at the end of the 19th century led to different models for the structure of the atom. In 1913, Niels Bohr first discovered is that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element.His produced a new "Shell" atomic model by adapted
Rutherford's "Planetary" model of the atom it was called the Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons.The negatively charged electrons were held in orbit by their attraction to the positively charged nucleus.He suggested that each electron has a set amount of energy, depending on the distance between the electron and the nucleus. If the electron took in more energy, for example , by the atom being heated, it could jump to the next level out, but it would eventually lose this extra energy and fall back to its original spot.which introduced photons.
Through discoveries of the electron and radioactivity at the end of the 19th century led to different models for the structure of the atom. In 1913, Niels Bohr first discovered is that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element.His produced a new "Shell" atomic model by adapted
Rutherford's "Planetary" model of the atom it was called the Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons.The negatively charged electrons were held in orbit by their attraction to the positively charged nucleus.He suggested that each electron has a set amount of energy, depending on the distance between the electron and the nucleus. If the electron took in more energy, for example , by the atom being heated, it could jump to the next level out, but it would eventually lose this extra energy and fall back to its original spot.which introduced photons.
|
The differences between Rutherford's model and the Bohr's model of the atomic structure are that,Rutherford's model described the atom as having a very tiny nucleus compared to the rest of the atom, where all the positive charge and most of the mass of the time was stored. The electrons were orbiting the nucleus just like planets orbit the sun. On the other hand Bohr's model is very similar to Rutherford's, except that in his model he uses quantum mechanics to explain the emission spectra of the various elements. In his model, the electrons are still orbiting the nucleus in circular paths.
|
M.F:
1. The electron in a hydrogen atom travels around the nucleus in a circular orbit. 2. The energy of the electron in an orbit is proportional to its distance from the nucleus. The further the electron is from the nucleus, the more energy it has. 3. Only a limited number of orbits with certain energies are allowed. In other words, the orbits are quantized. 4. The only orbits that are allowed are those for which the angular momentum of the electron is an integral multiple of Planck's constant divided by 2. 5. Light is absorbed when an electron jumps to a higher energy orbit and emitted when an electron falls into a lower energy orbit. 6. The energy of the light emitted or absorbed is exactly equal to the difference between the energies of the orbits. |