James Chadwick
|
R.V:James Chadwick was an English physicist who was awarded the 1935 Nobel Prize in Physics for his discovery of the neutron in 1932.he was born in Cheshire, England, on 20th October, 1891 and died on July 24, 1974 at Cambridge in Cambridgeshire.
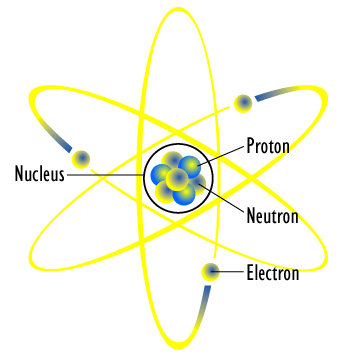
In 1921, Rutherford suggested that a further particle might exist.It would be roughly the same size as a proton, but without a charge, which he called the neutron.James Chadwick discovered the neutron using evidence collected by Irene Joliot-Curie, who discovered that when beryllium was bombarded with positively charged alpha particles a beam with a high penetrating power was created.Chadwick discovered that this beam was not deflected by either electric or magnetic fields, showing that it contained neutral particles called neutrons. It wasn’t until 1932 that James Chadwick was able to show that neutrons did exist. Chadwick was later awarded a Nobel prize in physics for his discovery.Neutrons are located in the centre of an atom, in the nucleus along with the protons. They have neither a positive nor negative charge, but contribute the the atomic weight with the same effect as a proton in the Bohr atomic structure model. The differences between Chadwick’s and Bohr’s atomic model.Bohr's model depicted the atom as a small, positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with electrostatic forces providing attraction, rather than gravity.However Chadwick discovered the neutron. Chadwick’s discovery made it possible to create elements heavier than uranium in the laboratory, and paved the way to uncovering the mechanism behind nuclear fission. |
M.F:James Chadwick conducted a series of experiments that would change the world of science forever. Although we now know that there are protons, neutrons, and electrons in an atom. In Chadwick's time most of this was still being experimented upon. In the beginning the experiments he did were to prove that if alpha particles fell on light elements such as beryllium, boron,and lithium gamma radiation would not be the result. Chadwick's hypothesis was correct, it wasn't gamma radiation but rather a beam of neutrons.
|